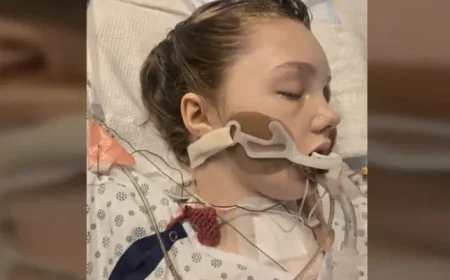
FDA Warns Novo Nordisk About Ozempic and Wegovy Side Effects

The U.S. Food and Drug Administration (FDA) has issued a warning to Novo Nordisk concerning its diabetes medications Ozempic and Wegovy. The warning centers around unreported side effects associated with these drugs.
FDA’s Warning Details
The FDA’s warning highlights concerns about safety signals that were not disclosed by the company. These signals are critical for patient safety and must be communicated effectively.
Impacted Medications
- Ozempic: A medication primarily used to manage type 2 diabetes.
- Wegovy: Designed for weight management in individuals with obesity.
Regulatory Oversight
The FDA’s scrutiny of Novo Nordisk is part of larger regulatory efforts to ensure that pharmaceutical companies report safety data accurately. This decision reflects a growing focus on transparency, especially in the wake of rising concerns over medication side effects.
Significance of the Warning
As one of the largest employers in the Triangle region, Novo Nordisk’s operations are under heightened observation. The company must address these safety concerns to maintain public trust and comply with regulatory standards.
Looking Ahead
Novo Nordisk’s response to the FDA will be critical in determining the next steps. The ongoing dialogue between the company and regulatory authorities will shape the future of Ozempic and Wegovy.
This warning serves as an important reminder of the need for diligence and transparency in the pharmaceutical industry, particularly regarding the safety of medications that millions rely on for their health.