Eye Drop Recall Raises a Bigger Question About What Consumers Were Not Told
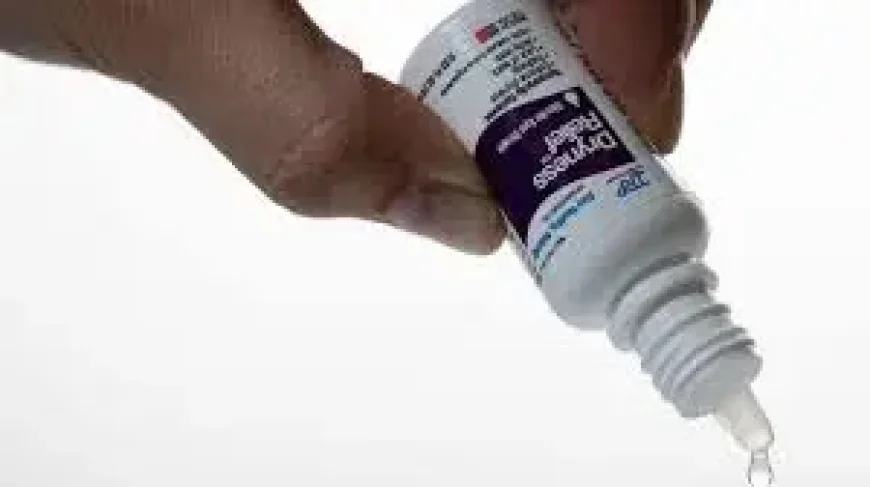
The recall of more than 3. 1 million eye drop products is large enough to change how consumers look at a routine item they may trust without a second thought. In the case of eye drop products, the scale alone is the first warning sign: a nationwide action involving millions of bottles suggests a problem that reaches far beyond a single shelf or store.
What is the recall actually telling consumers?
Verified fact: The context identifies a recall impacting over 3. 1 million eye drop products distributed nationwide and ties that action to the Food and Drug Administration. That is the clearest documented point in the record. The headline framing also makes one thing plain: this is not a narrow, isolated event. It is a broad recall with national distribution.
Informed analysis: When a product used so routinely enters recall territory at this scale, the central public question is not only what was affected, but how many people may already have it in homes, workplaces, or medical settings. The broader concern is whether consumers received enough notice to identify the exact products in time. For eye drop buyers, the practical issue is immediate: which bottles are included, and which are not?
How wide is the distribution problem?
The most significant detail in the context is the number itself: more than 3. 1 million eye drop products distributed nationwide. That volume signals a reach that is hard to dismiss as a limited quality issue. It also means the recall is not confined to one location or one narrow channel of sale.
Verified fact: The recall is national in scope. Informed analysis: National distribution creates a second layer of risk because consumers may not remember where a product was purchased, when it was bought, or whether it came from one of many affected lots. That is why recall clarity matters: if the warning is vague, the remedy becomes difficult to carry out. In a recall of eye drop products, precision is not a technical detail; it is the only way the public can act on the warning.
That is also where the public interest sharpens. A recall of this size implies that a large number of people may need to check their medicine cabinets, not just a small subset of users. The difference between a clear recall and an incomplete one is whether consumers can quickly separate safe products from recalled eye drop products.
Who is involved, and who is left with the burden?
The named institution in the context is the Food and Drug Administration. That makes the recall an official consumer-safety matter, not a rumor or a private warning. The recall itself is the public-facing action; the burden of response, however, falls on consumers who must identify the product and remove it from use.
Verified fact: The FDA is the institution connected to the recall. Informed analysis: That matters because official action gives the public a basis for trust, but trust alone does not solve the practical problem. Consumers still need clear product identifiers, and the context provided here does not include the exact product names or affected lots. That absence is important. It means the public cannot be expected to rely on memory alone when dealing with eye drop products that may look similar on a shelf.
Who benefits from clarity? Consumers do, because they can quickly determine whether they are exposed. Who is burdened by uncertainty? Consumers do, because uncertainty forces them to pause use, check packaging, and wait for more detailed identification. In a recall of this size, lack of detail is not neutral. It pushes the work of verification onto the public.
What should the public understand now?
The headline and context together point to a simple but serious takeaway: a nationwide recall affecting over 3. 1 million eye drop products deserves close attention even when the available record is limited. The fact pattern is narrow, but the implication is broad. Millions of units are involved, distribution is nationwide, and the official institution linked to the action is the FDA.
Verified fact: More than 3. 1 million eye drop products are affected. Informed analysis: That number alone suggests why consumers should not treat this as a routine alert. Eye drop products are typically used with a presumption of safety and precision. A recall at this scale interrupts that presumption. The public should want immediate, product-specific clarity, because a recall without specifics leaves room for confusion, delay, and unnecessary exposure.
For now, the evidence in the record supports restraint rather than speculation. The facts are clear on scope and official involvement, but the context does not supply the product list, the reason for the recall, or the exact distribution chain. That absence is itself part of the story: the public is being asked to pay attention before the full picture is visible.
That is why the eye drop recall matters beyond the headline. It is not only about one product category; it is about whether consumers can get enough information to protect themselves quickly, accurately, and without guesswork. Until that clarity is available, the scale of the recall remains the most important fact in the case of eye drop products.