Targeted In Vivo Engineering Reprograms T Cells
Recent advancements in T cell engineering continue to demonstrate promising potential in immunotherapy. Specifically, in vivo engineering techniques for reprogramming T cells are gaining attention. This article explores vital methodologies and findings related to targeted in vivo engineering of T cells.
Sources of Donor Cells
The study utilized leukapheresis products sourced from de-identified healthy donors, with ethical oversight verified through Institutional Review Board (IRB)-approved consent protocols from StemCell Technologies and Vitalant.
Animal Model Protocols
All experimental procedures involving animals adhered to protocols approved by the UCSF Institutional Animal Care and Use Committee (IACUC). This included the acknowledgment of humane endpoints in various experimental settings.
Plasmid Information for T Cell Engineering
- Transient GFP expression used the scAAV-CAG-GFP plasmid.
- sgRNA targeted the TRAC locus for gene integrations.
- Construction of CARs utilized various sequences including BCMA-targeting domains.
Cell Culture Conditions
Several cell lines essential for this research were cultured under controlled conditions. HEK293T cells were maintained in GlutaMAX DMEM and supplemented with fetal bovine serum (FBS) and antibiotics. Various T cell lines, such as NALM6 and OPM2, were cultured in RPMI 1640 with similar supplements.
Methods of Human T Cell Isolation
- PBMCs were utilized directly or frozen.
- T cells were activated with Dynabeads and cultured in X-VIVO medium supplemented with specific cytokines.
Engineering Techniques
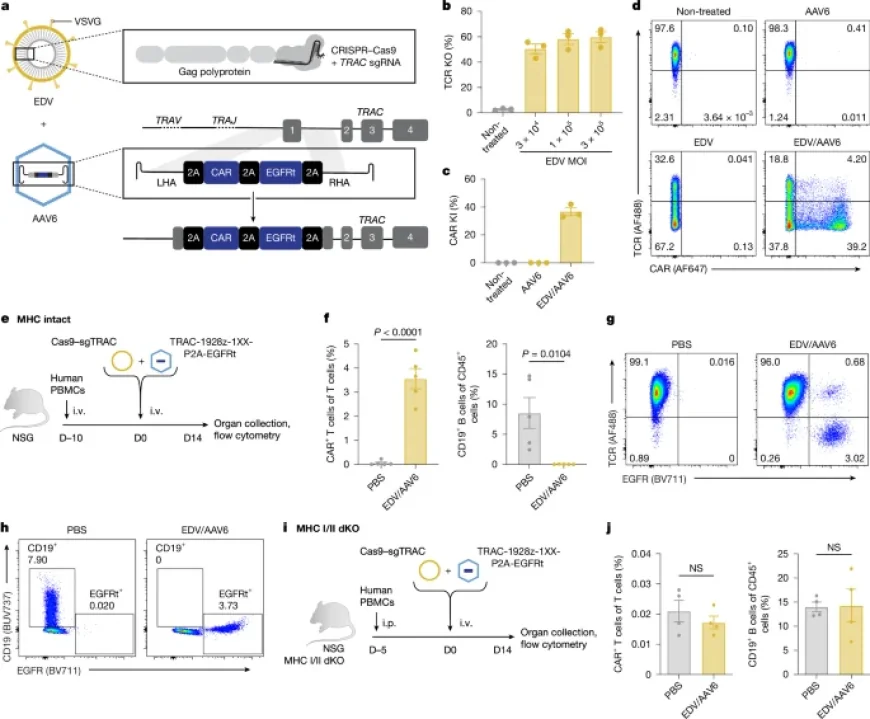
The study primarily focused on engineered nucleases such as Cas9, combined with delivery systems like envelope-decorated vehicles (EDVs). These systems enhance the introduction of specific genetic modifications into T cells.
Viral Vector Usage
- Second-generation lentiviruses were used for CAR delivery.
- Adeno-associated viruses (AAVs) facilitated gene transfer using capsid libraries.
Quality Control and Quantification
Post-transfection analyses included screening for viral efficacy and quantifying sgRNA concentrations through RT-qPCR methods. Outputs were analyzed to ensure the targeted integration of keywords and sequences.
Animal Models for In Vivo Studies
A range of murine models, specifically NSG and NSG-MHCI/II double-KO mice, were used for in vivo experiments. These models were essential for validating T cell performance and tumor suppression capabilities.
Humanization Procedures
Mice underwent humanization using healthy donor PBMCs. Following an integration phase, various T cell injections were administered to assess in vivo efficacy.
Biodistribution and Cytokine Profiling
Biodistribution studies assessed the localization of engineered T cells and their associated cytokines post-administration. Serum samples were collected and analyzed to profile immune responses generated by the treatments.
Analysis Procedures
- Genomic DNA from isolated CAR T cells was sequenced to characterize TCR clonality.
- Flow cytometry was employed to evaluate T cell populations and performance metrics.
This study underlines the complexities and advancements in targeted in vivo engineering of T cells, highlighting innovative methodologies and their significant implications in the field of immunotherapy.