Bayer Orders Afrin Recall of 786,100 Travel-Size Bottles
Bayer issued an afrin recall on Thursday for 786,100 units of Afrin Original Nasal Spray in 6 mL travel-size bottles after the packaging was found to be not child resistant. The U.S. Consumer Product Safety Commission announced the recall the same day and said customers who bought the product can get a refund.
CPSC notice on Afrin bottles
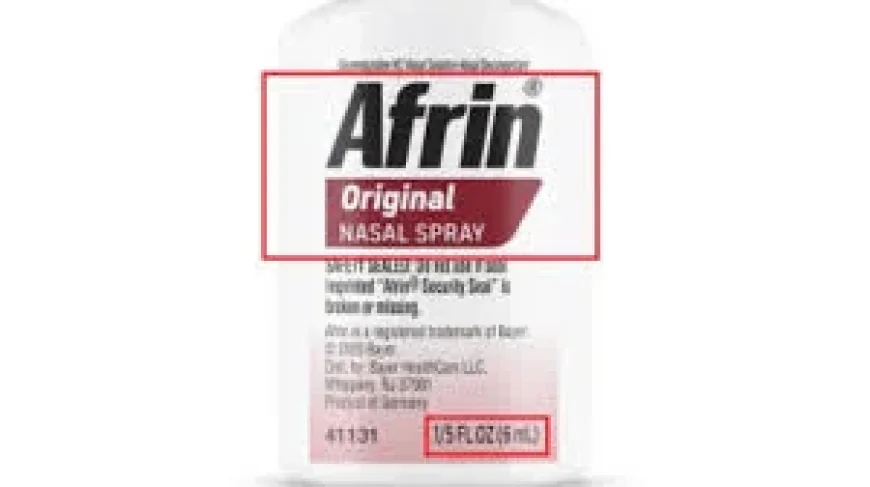
The recall covers unexpired Travel Size Afrin Original Nasal Spray 6 mL bottles with lot numbers 230361, 240822, 241198, 250066, 250152, 250646, and 250831. The bottles have “Afrin Original Nasal Spray” and “1/5 FL OZ (6 mL)” printed on a label on the front.
The consumer safety notice said the 6 mL nasal spray’s packaging is not child-resistant and does not bear the required labeling statement. It said that creates a risk of serious injury or illness from poisoning if young children swallow the contents.
Refunds for affected buyers
The recall affects a nationwide batch of travel-size Afrin bottles sold nationwide, putting the packaging itself at the center of the action rather than the medicine inside. No injuries have been reported in connection with the recall, but the notice still directs customers who purchased the product to seek a refund.
For buyers, the practical step is straightforward: check the bottle’s front label and lot number against the numbers listed in the notice. If the bottle matches one of the recalled lots, it falls within the refund offer announced by the consumer safety commission.
Travel-Size Afrin lot numbers
Lot numbers in the recall are 230361, 240822, 241198, 250066, 250152, 250646, and 250831. The affected product is the 6 mL travel-size version of Afrin Original Nasal Spray.