RFK Jr. Promises Action on Peptides, Exciting Wellness Community
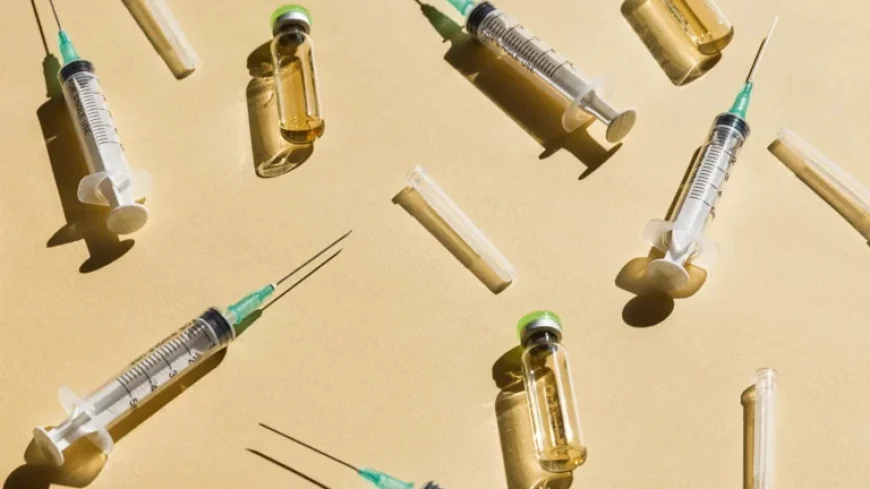
Synthetic peptides are emerging as a popular option for wellness and longevity, despite safety concerns and a lack of comprehensive human testing. Health Secretary Robert F. Kennedy Jr. recently indicated that the federal government might soon ease restrictions on certain peptides. His comments, made during an appearance on the Joe Rogan podcast, suggest that the FDA could approve around 14 synthetic peptides within a few weeks, allowing ethical suppliers to offer them to consumers.
Understanding Synthetic Peptides
Peptides are short chains of amino acids that occur naturally in the body. Interest in their synthetic counterparts is increasing, driven by claims of benefits for tissue repair, skin health, and longevity. However, these synthetic peptides are currently not approved for use by the FDA, which restricts their compounding by pharmacies due to safety concerns.
Current Regulatory Landscape
The FDA has not permitted the compounding of popular synthetic peptides such as BPC-157 and ipamorelin. These restrictions are in place primarily due to a lack of safety and efficacy data from human trials. Emily Hilliard, a spokesperson for the Department of Health and Human Services, emphasized that the FDA aims to ensure consumer access to safe products produced under rigorous quality standards.
The Risks of an Unregulated Market
- Increasing black market for peptides.
- Potential dangers from untested compounds.
- Injecting peptides sourced from unreliable suppliers.
Experts like Dr. Myles Spar caution against using unapproved peptides, as they might pose health risks. He believes that regulatory changes could provide patients with safer access and mitigate the dangers associated with unregulated sources. Scott Brunner, CEO of the Alliance for Pharmacy Compounding, supports the idea that the FDA should allow compounding of peptides with established safety records.
Importance of Clinical Testing
Despite the growing interest in peptide therapies, many of these compounds lack the necessary clinical trial data proving their safety and efficacy. Eileen Kennedy, a chemical biologist, warns that even if peptides are naturally occurring, injecting them in higher doses could produce unforeseen harmful effects. Clinicians advocating for peptide use argue that waiting for extensive human trials may hinder potential advancements in medicine.
Challenges in Regulation and Approval
Dr. Edwin Lee, an endocrinologist involved in peptide education, suggests that the lack of patentability for many peptides discourages pharmaceutical companies from investing in the research required for formal FDA approval. This has left many practitioners operating within an unregulated landscape, compounding their own peptides from suppliers that may not meet the stringent standards expected by the FDA.
Future of Peptide Therapies
- Potential for FDA to approve more peptides in subsequent months.
- Need for compounding pharmacies to adapt to new regulations.
- Concerns about the safety of unregulated products persist.
Even if the FDA approves certain peptides for compounding, a delay may occur as pharmacies gear up with the necessary supplies. Legal experts suggest that transferring these synthetic peptides from an unregulated market into a controlled environment is a step in the right direction. Monitoring consumers effectively will be crucial to avoid the rise of black market products. As this situation evolves, one thing remains clear: the intersection of synthetic peptides, health, and regulatory action is set to shape the future of wellness treatments.